Blue Button
The Blue Button symbol signifies that a site has functionality for customers to download health records. You can use your health data to improve your health and to have more control over your personal health information and your family’s healthcare. Look for the Blue Button symbol and take action using your personal health information.

Blue Button may be able to help you take action using your personal health information.
- Do you want to feel more in control of your health and your personal health information?
- Do you have a health issue?
- Are you caring for an elderly parent?
- Are you changing doctors?
- Do you need to find the results of a medical test or a complete and current list of your medications?
Your Health Records
Health information about you may be stored in many places, such as doctors’ offices, hospitals, drug stores, and health insurance companies. The Blue Button symbol signifies that an organization has a way for you to access your health records electronically so you can:
- Share them with your doctor or trusted family members or caregivers
- Check to make sure the information, such as your medication list, is accurate and complete
- Keep track of when your child had his/her last vaccination
- Have your medical history available in case of emergency, when traveling, seeking a second opinion, or switching health insurance companies
- Plug your health information into apps and tools that help you set and reach personalized health goals.
You have a legal right to receive your personal health information. Blue Button is one of the ways this information may be made available to you. Look for the Blue Button symbol, and ask your health care providers or health insurance company if they offer you the ability to view online, download, and share your health records.
Your Rights
As Americans, we each have the legal right to access our own health records held by doctors, hospitals and others who provide healthcare services for us. And we have the option of getting our records on paper or electronically depending on how they are stored. You can exercise your rights by downloading your health records through an online portal, or by asking how to get a copy of your health records. Some doctors or hospitals may not be familiar with your rights to access your information about your own health. For more information about your health information rights, how to share your health information, and how to protect it, please see the Your Rights Infographic.
Download Infographic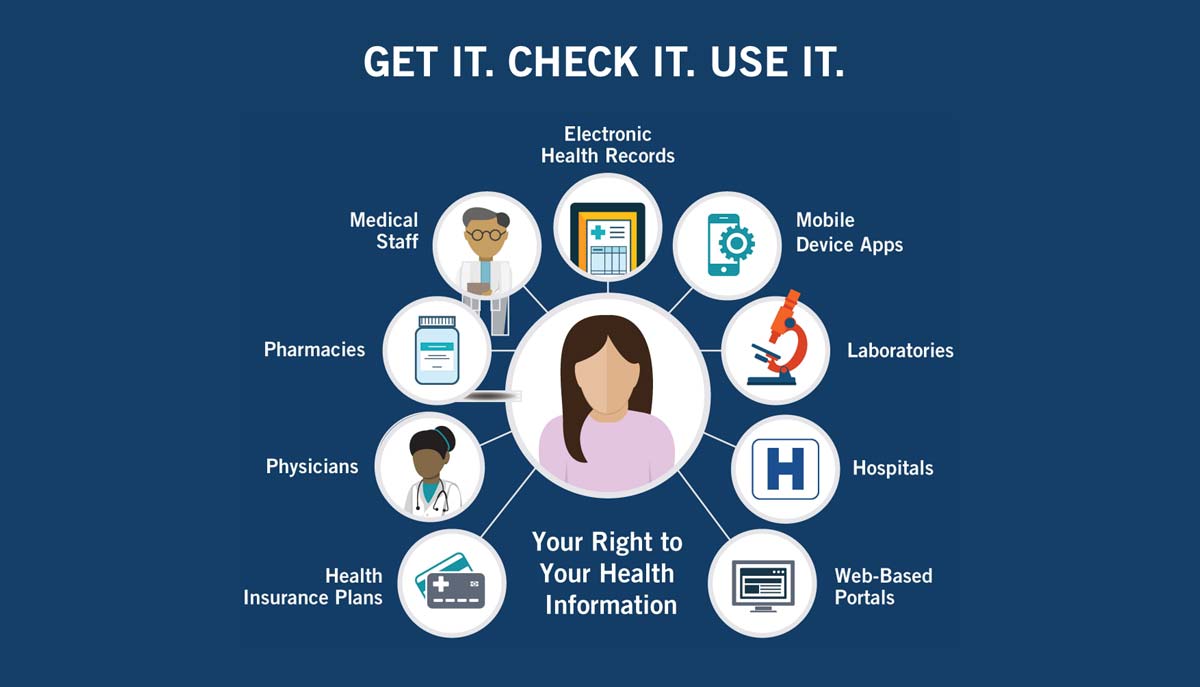
What kind of information is available to you?
It depends on whether you are getting information from your health care provider (doctor, hospital, nursing home, etc.), your health insurance company, or another source such as a drug store or a lab since each has different kinds of information. In general, you may expect to be able to electronically access important information such as:
- Current medications you are taking
- Any allergies you have
- Medical treatment information from your doctor or hospital visits
- Your lab test results
- Your health insurance claims information (financial information, clinical information and more)
Until recently, many health records were stored in paper files, so it wasn’t very easy for you to access or use this information. But that is changing as more doctors and hospitals adopt electronic health records (EHRs) and other health information technologies, including mobile health apps.
Medicare beneficiaries can view and download their Medicare claims data in a more timely and user-friendly format than ever before. That information now covers three years of your health history, including claims information on services covered under Medicare Parts A and B, and a list of medications that were purchased under Part D. Look for the Blue Button symbol on the MyMedicare web site.
Veterans can find the Blue Button symbol on the MyHealtheVet website and download demographic information (age, gender, ethnicity and more), emergency contacts, a list of their prescription medications, clinical notes, and wellness reminders.
You may want to check back often as more and more organizations join the Blue Button movement. Online health records are not yet available to everyone, but access is rapidly growing, and if you ask for access you can help grow it faster.
Frequently Asked Questions
Blue Button Logo and Usage
The words “Blue Button,” the Blue Button logo, the Blue Button combined logo and the slogan “Download My Data” are what consumers look for when they want electronic access to their health data. You are free to use these service marks as long as you follow the guidelines and service mark use rules below.
- First, the Blue Button marks are to be used to identify electronic access to health data views, downloads, and transmissions. Health data includes clinical, demographic, financial (explanation of benefits or invoices) and other information related to the health and medical care of a patient.
Use of the Blue Button marks for any other types of data, information, or for any other purpose is not permitted. - Second, patients should be able to read their Blue Button information.
Machine-readable formats are fine, so long as humans can easily read and print them (e.g. through a freely available viewer). - Third, the Blue Button marks may not be used in any manner that could give rise to the appearance of endorsement or solicitation on behalf of the Department of Health and Human Services or the U.S. government. A non-endorsement statement must be included if the Blue Button marks are used with another trademark or in an advertisement for other products and services. An example of a non-endorsement statement is “Participation by [XYZ Co.] does not imply endorsement by HHS or the U.S. government.”
It’s your product or service. Don’t suggest in any way that the federal government or any of its agencies endorse or support your product or service. - Finally, the Blue Button marks are registered trademarks. Proper use of the Blue Button marks with the trademark symbol lets everyone know they are legally registered and protected by trademark law.
Use the ® symbol to designate the registered status of the Blue Button marks.
- Use the ® symbol the first time you use the words “Blue Button” or “Download My Data” in a document or on a page, and make sure the ® appears any time you use one of the graphic marks. The ® is included in the graphics files we’ve made available elsewhere on this page.
- The marks should be used and displayed in their entirety, without variation in design or color, and without other elements superimposed on them. If you’re not printing in color, grey tones can be used instead of blue.
- Use the following attribution statement nearby — at least on the first page of the use of any of the marks, or on a “notice” page: “Blue Button, the slogan, ‘Download My Data,’ the Blue Button Logo, and the Blue Button combined logo are registered service marks owned by the U.S. Department of Health and Human Services.”
- When referring to the marks in text always use the mark as an adjective, not a noun. For example, the Blue Button Electronic Health Record Retrieval System.
- Should you become aware of any misuse of the marks, tell us by sending an email to: BlueButtonMarks@hhs.gov. Please include details of the alleged misuse, such as screen shots and copies of the improper use.
- Q: Who owns the Blue Button marks?
A: The words “Blue Button,” the Blue Button logo, the Blue Button combined logo, and the slogan, “Download My Data” are registered service marks of the U.S. Department of Health and Human Services. - Q: Why are the Blue Button marks trademarked?
A: The federal government registered the Blue Button marks to protect their meaning and use, and to provide a way for others who support the Blue Button principles, particularly individual consumer or patient electronic access to their own health information, in a format they can use. - Q: Do I need a written license to use the Blue Button marks?
A: A written license is not required to use the Blue Button marks. As long as the guidelines and the Service Mark Use Rules are followed, the Blue Button marks are available for your use. By using the Blue Button marks, you accept and agree to follow these guidelines and rules. - Q. What are the Blue Button Marks?
A: There are four registered service marks and they must be used as shown below.- Standard Word Mark: Blue Button
- Slogan Word Mark: Download My Data
- Blue Button logo:

- Combined logo:

- Q: Where can I get graphics files of the Blue Button marks?
A: Download an entire library of high- and screen-resolution marks [ZIP – 1.9 MB]. - Q: The guidelines say that if I use the Blue Button Marks, I must acknowledge them as service marks. What does that mean and what do I have to do?
A: A few simple but important items to remember:- Use the marks as shown above only to identify health data views, downloads and transmissions; and
- Acknowledge that they are owned by the U.S. Department of Health and Human Services (HHS). Any other use could create confusion for consumers. Also, follow the service mark use rules above.

